About dystrophic epidermolysis bullosa (DEB)
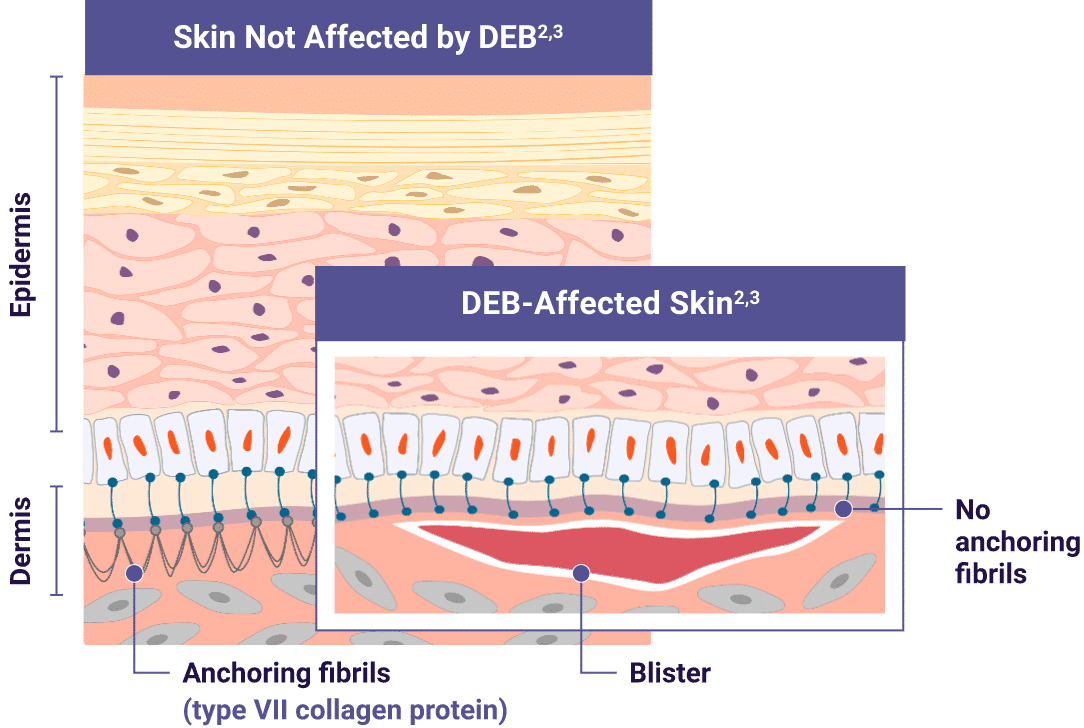
DEB is a disease of skin fragility caused by mutations in the gene COL7A1 1
Adapted from De Rosa L, Latella MC, Secone Seconetti A, et al.
Cold Spring Harb Perspect Biol. 2020.
- These mutations in the COL7A1 gene result in the lack of functional type VII collagen, which disrupts the formation of anchoring fibrils in the skin and prevents adhesion of the epidermis to the dermis1
- There are 2 types of DEB based on inheritance patterns: dominant (DDEB) and recessive (RDEB)4
- DDEB results in reduced Type VII collagen protein levels
- RDEB results in absent or markedly reduced Type VII collagen protein levels
- Regardless of type, patients and families can face significant physical, emotional, and financial burdens1
Genetic testing can confirm a DEB diagnosis.5
Learn more about genetic testing for DEB.
Wounds that heal without Type VII collagen are either chronic or reopen easily, which may lead to serious complications6
Inflammation
Deformities
Cell Carcinoma (SCC)
A lack of Type VII collagen can cause lifelong blistering and slow wound healing, leading to wounds prone to bacterial infections, scarring, and inflammation. Over time, chronic inflammation and the constant reopening of wounds or chronic wounds increase the risk of developing SCC.6,7
Learn about a treatment option
See How It WorksVYJUVEK is a topical gel indicated for the treatment of wounds in adult and pediatric patients (from birth) with dystrophic epidermolysis bullosa (DEB) with mutation(s) in the collagen type VII alpha 1 chain (COL7A1) gene.
WARNINGS AND PRECAUTIONS
Accidental Exposure to VYJUVEK gel: VYJUVEK will not replicate in the subject’s cells and does not integrate into the subject cells’ native genetic material. For precautions, avoid direct contact with treated wounds (e.g., touching or scratching) and dressings of treated wounds until the next dressing change following treatment. Wear protective gloves when assisting patients with changing wound dressings and handling the disposal. In the event of accidental exposure (e.g., through a splash to the eyes or mucous membranes), flush with clean water for at least 15 minutes.
Clean all surfaces that may have come in contact with VYJUVEK biological suspension or gel and treat all spills with a virucidal agent.
Dispose all materials (e.g., vial, syringe, cleaning materials) that may have come in contact with VYJUVEK biological suspension or gel into a biohazard container.
ADVERSE REACTIONSThe most common adverse reactions (>5%) were itching, chills, redness, rash, cough, and runny nose.
- Please see Important Safety Information above and click here for full Prescribing Information.
1. Paller AS, Guide SV, Deigo A, et al. Practical considerations relevant to treatment with the gene therapy beremagene geperpavec-svdt for dystrophic epidermolysis bullosa. J Dermatolog Treat. 2024;35(1):2350232. doi:10.1080/09546634.2024.2350232
2. De Rosa L, Latella MC, Secone Seconetti A, et al. Toward Combined Cell and Gene Therapy for Genodermatoses. Cold Spring Harb Perspect Biol. 2020;12(5):a035667. doi:10.1101/cshperspect.a035667
3. Eichstadt S, Barriga M, Ponakala A, et al. Phase 1/2a clinical trial of gene-corrected autologous cell therapy for recessive dystrophic epidermolysis bullosa. JCI Insight. 2019;4(19):e130554. doi:10.1172/jci.insight.130554
4. Denyer J, Pillay E, Clapham J. Best Practice Guidelines for Skin and Wound Care in Epidermolysis Bullosa. An International Consensus. Wounds International; 2017.
5. Has C, Liu L, Bolling MC, et al. Clinical practice guidelines for laboratory diagnosis of epidermolysis bullosa. Br J Dermatol. 2020;182(3):574-592.
6. Bardhan A, Bruckner-Tuderman L, Chapple ILC, et al. Epidermolysis bullosa. Nat Rev Dis Prim. 2020;6:78.
7. Condorelli AG, Dellambra E, Logli E, et al. Epidermolysis bullosa—associated squamous cell carcinoma: from pathogenesis to therapeutic perspectives. Int J Mol Sci. 2019;20(22):5707.
VYJUVEK is a topical gel indicated for the treatment of wounds in adult and pediatric patients (from birth) with dystrophic epidermolysis bullosa (DEB) with mutation(s) in the collagen type VII alpha 1 chain (COL7A1) gene.
WARNINGS AND PRECAUTIONS
Accidental Exposure to VYJUVEK gel: VYJUVEK will not replicate in the subject’s cells and does not integrate into the subject cells’ native genetic material. For precautions, avoid direct contact with treated wounds (e.g., touching or scratching) and dressings of treated wounds until the next dressing change following treatment. Wear protective gloves when assisting patients with changing wound dressings and handling the disposal. In the event of accidental exposure (e.g., through a splash to the eyes or mucous membranes), flush with clean water for at least 15 minutes.
Clean all surfaces that may have come in contact with VYJUVEK biological suspension or gel and treat all spills with a virucidal agent.
Dispose all materials (e.g., vial, syringe, cleaning materials) that may have come in contact with VYJUVEK biological suspension or gel into a biohazard container.
ADVERSE REACTIONSThe most common adverse reactions (>5%) were itching, chills, redness, rash, cough, and runny nose.
- Please see Important Safety Information above and click here for full Prescribing Information.
1. Paller AS, Guide SV, Deigo A, et al. Practical considerations relevant to treatment with the gene therapy beremagene geperpavec-svdt for dystrophic epidermolysis bullosa. J Dermatolog Treat. 2024;35(1):2350232. doi:10.1080/09546634.2024.2350232
2. De Rosa L, Latella MC, Secone Seconetti A, et al. Toward Combined Cell and Gene Therapy for Genodermatoses. Cold Spring Harb Perspect Biol. 2020;12(5):a035667. doi:10.1101/cshperspect.a035667
3. Eichstadt S, Barriga M, Ponakala A, et al. Phase 1/2a clinical trial of gene-corrected autologous cell therapy for recessive dystrophic epidermolysis bullosa. JCI Insight. 2019;4(19):e130554. doi:10.1172/jci.insight.130554
4. Denyer J, Pillay E, Clapham J. Best Practice Guidelines for Skin and Wound Care in Epidermolysis Bullosa. An International Consensus. Wounds International; 2017.
5. Has C, Liu L, Bolling MC, et al. Clinical practice guidelines for laboratory diagnosis of epidermolysis bullosa. Br J Dermatol. 2020;182(3):574-592.
6. Bardhan A, Bruckner-Tuderman L, Chapple ILC, et al. Epidermolysis bullosa. Nat Rev Dis Prim. 2020;6:78.
7. Condorelli AG, Dellambra E, Logli E, et al. Epidermolysis bullosa—associated squamous cell carcinoma: from pathogenesis to therapeutic perspectives. Int J Mol Sci. 2019;20(22):5707.
For US Healthcare Professionals Only
This website is intended for US healthcare professionals.
Would you like to continue?
